Resources for each step of the treatment journey with
AUSTEDO XR
Downloadable resources
Fill out this form to enroll your patient in Teva Total Support™ (for patients taking AUSTEDO XR).
Fill out this form to enroll your patient in Teva Total Support™ (for patients taking AUSTEDO BID).
Use this template to write an appeals letter, which payers may need in order to appeal a denial of coverage for AUSTEDO XR or AUSTEDO tablets.
Use this template to write a letter to establish medical necessity, which payers may require for treatment with AUSTEDO XR or AUSTEDO.
This brochure provides an overview of Teva Total Support for your patients, including information about navigating insurance, financial assistance options, and enrollment.
Use this manual when prescribing AUSTEDO XR in the PointClickCare platform.
A guide with access and affordability information to get patients started on AUSTEDO XR.
This flashcard outlines improvements to Medicare made by the Inflation Reduction Act (IRA) to expand benefits and improve drug costs for many patients.
Accessing AUSTEDO XR is easy with Teva's broad specialty pharmacy network. Find the most up-to-date list of national and regional specialty pharmacies.
Downloadable resources for your patients with HD chorea
This brochure provides answers to important questions your patients with HD chorea and their care partners may have when starting treatment.
A useful resource for patients as they begin their treatment journey. Patients and their care partners can use this tracking tool to record when they take their treatment, as well as any changes in their HD chorea.
This resource is for patients to use at their next appointment with you—whether in person or through telemedicine—to help decide if AUSTEDO XR is right for them.
A resource introducing patients to Teva Total Support—a patient support partner that can help when getting started with AUSTEDO XR, and with finding financial assistance offerings to stay on treatment.
Videos to help you support your patients with HD chorea
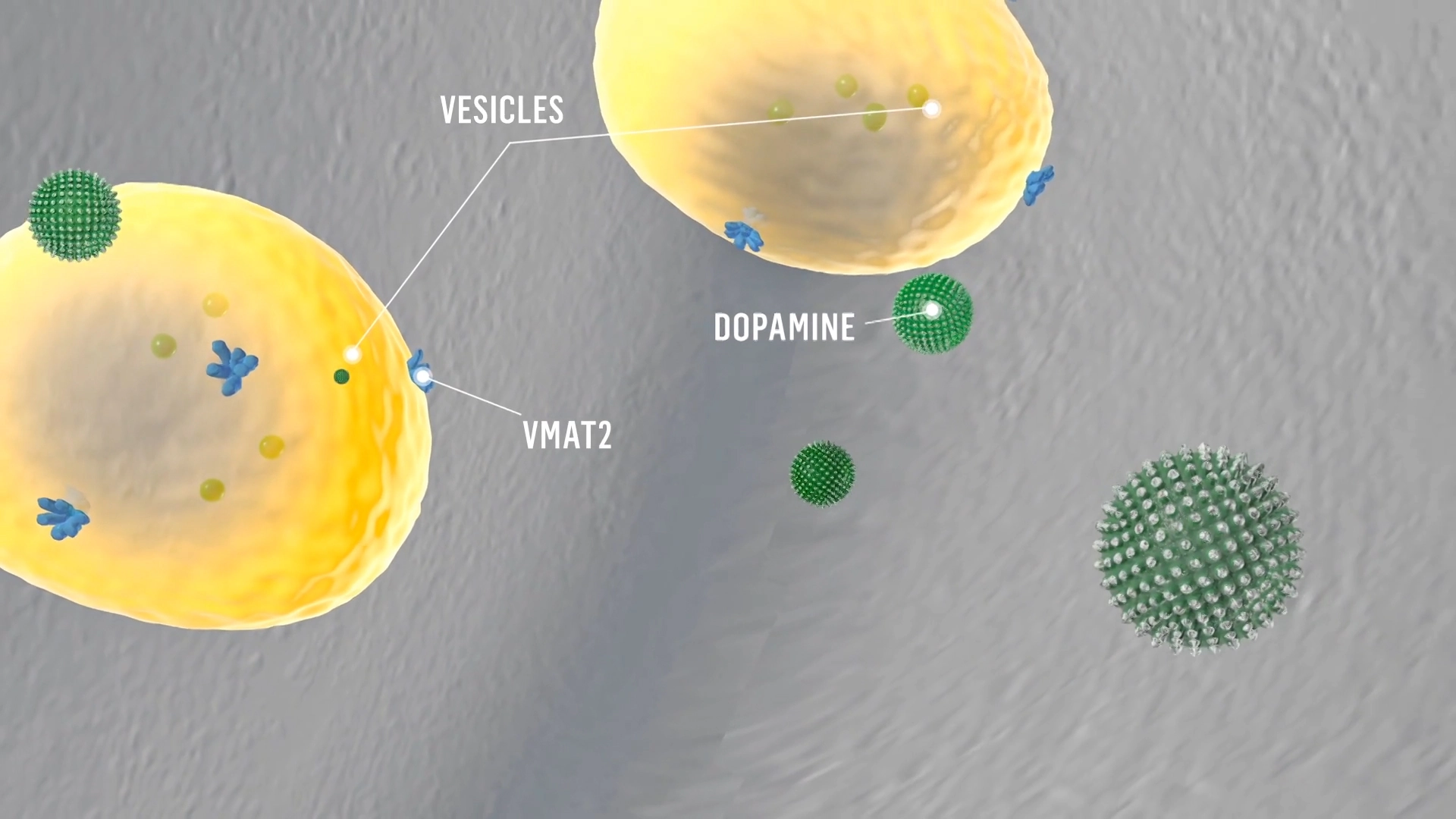
Mechanism of Action
VMAT2 inhibition can help regulate dopamine function.1
Voiceover:
Maintaining a balance in the level of dopamine is essential for controlled movement.
Dopamine signaling is facilitated by vesicular monoamine transporter 2, or VMAT2, which transports monoamines, including dopamine, from the cytosol into synaptic vesicles, keeping them ready for subsequent release in response to an action potential.
When an action potential reaches the nerve terminal of the presynaptic neuron, dopamine is released from the synaptic vesicles into the synaptic cleft.
This dopamine binds to receptors on the postsynaptic neuron, thereby signaling movement.
Excess dopamine signaling manifests as abnormal involuntary movements.
AUSTEDO® (deutetrabenazine) is a VMAT2 inhibitor.
Deutetrabenazine contains deuterium, a naturally occurring heavy version of hydrogen. Deuterium forms stronger molecular bonds with carbon atoms, thereby extending the half-life of therapeutic metabolites.
The precise mechanism by which deutetrabenazine exerts its effects on abnormal involuntary movements is unknown. It is believed to be related to its effect as a reversible depleter of monoamines, including dopamine, from nerve terminals.
Deutetrabenazine binds to VMAT2 on the vesicle in the presynaptic neuron and inhibits the uptake of dopamine into synaptic vesicles.
Dopamine molecules collect outside the blocked VMAT2 and are degraded by monoamine oxidase.
Reducing dopamine levels in the presynaptic neuron results in less dopamine signaling to the postsynaptic neuron.
Limiting dopamine signaling is believed to lead to fewer abnormal involuntary movements.

The Impact of HD Chorea
Eileen discusses how her life, and in particular, her work, has been affected by HD chorea.
Onscreen text:
A patient talks about how she had to stop work as a legal secretary due to her Huntington’s disease.
Eileen:
Well, it affected me and my working.
I’m not working anymore, I’m on disability, and I, uh…
It’s…’cause I can’t read as much, like I used to read. Um…
Rajeev Kumar:
What kind of work did you used to do?
Eileen:
I was a legal secretary.
Rajeev Kumar:
And how did it interfere with reading and doing your job?
Eileen:
It interfered with my reading pleadings and emails, um.
I can’t comprehend reading anymore. And I’ve been reading my whole life.
Um, I don’t read books like I used to.
In fact, I don’t read them at all.
And it has affected me in ways of eating.
I drop my food, I drop…I fall over. I fall on occasion.

Hear From an Expert: Pivotal Trial Results
Dr. Rajeev Kumar discusses SF-36 results and impact on activities of daily living.
Voiceover/Onscreen text:
INDICATIONS AND USAGE
AUSTEDO® XR (deutetrabenazine) extended-release tablets and AUSTEDO® (deutetrabenazine) tablets are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Rajeev Kumar:
Hello. I’m Dr. Rajeev Kumar, Medical Director of the Rocky Mountain Movement Disorders Center and Huntington’s Disease Society of America Center of Excellence in Colorado. For over 20 years, I have been working with patients to treat their chorea symptoms.
I was involved in the evaluation of AUSTEDO in the pivotal trial for treatment of chorea associated with HD. I chose to be involved because the success of this trial could lead to a treatment for my patients with chorea.
I had a conversation with a colleague the other day about our patients with HD chorea. AUSTEDO is an appropriate choice to treat those symptoms, and I thought it’d be insightful to share some of our discussion with you.
My colleague said he was concerned about the impact chorea symptoms were having on his patients and their caregivers. He was particularly concerned about patients who were not aware of their symptoms and had become increasingly reliant on their caregivers. Even when patients are not aware of their symptoms, it is still important to treat them because of the impact chorea can have on their ability to function and their need for daily assistance.
The efficacy and safety of AUSTEDO were demonstrated in a randomized, 12-week placebo-controlled study in patients with chorea. The primary endpoint was total maximal chorea, or TMC, score. AUSTEDO reduced TMC score by 4.4 points from baseline, more than twice the improvement in the TMC score from placebo. The reduction in chorea was demonstrated in the face, mouth, trunk, and legs.
In my experience, after patients begin treatment, they often acknowledge improvements in areas of self-care and activities of daily living. My colleague and I talked about how we have observed improvement in our patients first-hand—particularly in areas of daily living, including walking, bathing, dressing, lifting, carrying groceries, moderate to vigorous activities, or climbing stairs.
Both patients and clinicians reported treatment success as measured by the Patient Global Impression of Change, or PGIC, and Clinical Global Impression of Change, or CGIC. Treatment is considered successful when symptoms are rated as “much improved” or “very much improved.” On the PGIC, the patient success rate with AUSTEDO was 51 percent, compared with 20 percent for placebo.
The physical function portion of the Short-Form 36 Health Survey, known as the SF-36, supported these findings as well. In the clinical trial for AUSTEDO, there was a statistically significant reduction in disability and improvement in physical functioning compared with placebo at 12 weeks.
While we were talking about study results, I mentioned that I tell my patients the most common side effects can include drowsiness, dry mouth, diarrhea, and fatigue. And I encourage them to share with me any symptoms they find bothersome.
Disclaimer at bottom of screen:
Patients who participated in the clinical trials discussed in this testimonial received the BID formulation of AUSTEDO.
Rajeev Kumar:
In my practice, I have been pleased with the experience my patients with HD chorea have had with AUSTEDO, particularly with their improvements in areas of daily living, physical functioning, and reduced disability. In addition to managing symptoms, I am always looking for opportunities to offer my patients a choice. With the availability of AUSTEDO XR, my patients have the option to take their treatment once daily.
Voiceover/Onscreen text:
INDICATIONS AND USAGE
AUSTEDO® XR (deutetrabenazine) extended-release tablets and AUSTEDO® (deutetrabenazine) are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics, The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO. Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.

Hear From an Expert: ARC-HD Trial Results
Dr. Fahd Amjad discusses long-term results through 3 years.
Voiceover/Onscreen text:
INDICATIONS AND USAGE
AUSTEDO® XR (deutetrabenazine) extended-release tablets and AUSTEDO® (deutetrabenazine) tablets are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Fahd Amjad:
Hello. I am Dr. Fahd Amjad, Co-Director of the Huntington’s Disease Care, Education, Research Center at Georgetown University Medical Center in Washington, DC.
For over 10 years, I have led our mission to provide treatment to and manage the symptoms experienced by patients with Huntington’s disease, or HD.
I ran into a colleague the other day, and thought I would share the conversation we had about our patients with HD chorea. More than 90 percent of patients with HD have chorea, and as neurologists, we appreciate the challenges faced by patients with chorea symptoms.
We both regularly observe that patients with chorea have difficulty walking and may fall. My colleague recently started managing a new patient with chorea symptoms and wanted to understand what my goals are for patients. Because patients with chorea are at risk of falls, they often become dependent on their partners or caregivers for help with their daily activities. One of my goals is to help patients maintain their independence for as long as possible.
I asked my colleague what he prescribes for his patients with chorea. Both of us consider AUSTEDO, a vesicular monoamine transporter 2 inhibitor, or VMAT2, for the management of chorea symptoms in patients with HD.
I choose AUSTEDO because its efficacy and safety were demonstrated in a randomized, 12-week, placebo-controlled study in patients with chorea associated with HD. The results showed a statistically significant reduction in chorea of the face, mouth, trunk, arms, and legs as measured by the total maximal chorea score.
AUSTEDO is also the first VMAT2 inhibitor to have physicians and patients rate overall HD chorea symptoms as “much improved” or “very much improved” when compared to placebo as measured by the Clinical Global Impression of Change and Patient Global Impressions of Change scales.
I mentioned to him that I recently reviewed a 3-year open-label extension study called ARC-HD, published in CNS Drugs. This AUSTEDO study interests me as a provider because HD is a progressive condition, and the study demonstrated about 3 years of continued control of chorea in patients.
Disclaimer at bottom of page:
Patients who participated in the clinical trials discussed in this testimonial received the BID formulation for AUSTEDO.
Fahd Amjad:
I am also confident in the demonstrated safety profile of AUSTEDO and explain to my patients that the most common side effects include somnolence, diarrhea, dry mouth, and fatigue. These side effects occur most often during the dosing titration phase.
Most of my patients are on an average daily of 36 mg with some up to 48 mg, which aligns with the clinical trials for AUSTEDO. And I can also offer my patients more options with the introduction of AUSTEDO XR, a once-daily formulation.
Voiceover/Onscreen text:
INDICATIONS AND USAGE
AUSTEDO® XR (deutetrabenazine) extended-release tablets and AUSTEDO® (deutetrabenazine) tablets are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics, The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.

Moves That Matter: Ray & Rhonda’s Long-Term Journey With HD Chorea
See how sustained chorea control with AUSTEDO has helped Ray and his wife and caregiver, Rhonda.
Ray:
I’m Ray…I’m 55.
Rhonda:
My name is Rhonda, and I am Ray’s wife.
Disclaimer at bottom of screen:
This was an actual patient’s experience. Results may vary. Ray and Rhonda were compensated for their participation in this video.
Onscreen text:
Ray and Rhonda were looking forward to spending their retirement hiking, traveling, and enjoying their grandchildren.
But when Ray was diagnosed with HD in 2019, it seemed like chorea would make all of that impossible.
Rhonda:
Ray’s dad had Huntington’s disease…he wasn’t able to walk…he ended up in a wheelchair…
So that was kind of a really scary thought for me. I just assumed that our lives…would be over very soon as we knew it…
Onscreen text:
As Ray’s disease progressed, his chorea began to affect him day and night.
Rhonda:
So the first symptoms I noticed of his chorea was at night while he was sleeping. His arms would, you know, kind of start flailing and hitting and then kind of it would just be like his whole body…
Ray:
Yeah. When I was driving to work one day…and my hands are just shaking like this…
But yeah, it’s just weird, you know, having your hands move, no explanation…
Rhonda:
At that time, I was feeling a little bit desperate, like, something needs to change. We can’t do this anymore.
Onscreen text:
When Ray fell out of bed and dislocated his shoulder, Rhonda realized it was time to talk to Ray’s doctor.
Rhonda:
We made an appointment with the neurologist that week talking to her about how bad it had gotten.
And she said, well, I have this drug that I would like for you to try.
Onscreen text:
Ray’s doctor started him on AUSTEDO, and as his dose was titrated, Ray and Rhonda began noticing an improvement in Ray’s chorea.
More than 3 years later, Ray is still taking AUSTEDO at 48 mg/day. His chorea movements have remained minimal.
Disclaimer at bottom of screen:
Once-daily AUSTEDO XR is available.
Bioequivalence of once-daily AUSTEDO XR has been established with AUSTEDO BID based on pharmacokinetic profile studies.
When two formulations are shown to be bioequivalent, they are considered to be therapeutically equivalent.
Rhonda:
Yeah. So his movements just became less and less as…the doses increased…
His sleeping now is very different…no more kicks, no more punches, no more flailing around in bed.
And…we both noticed that the small things that he would do with his hands became easier.
Ray:
Yeah.
Rhonda:
Buttoning his shirt, putting the leash on the dog’s collar, tying shoes. Those things became easier.
So yeah…it has far exceeded our expectations…
Onscreen text:
Today, Ray and Rhonda are still on the move—enjoying an active lifestyle and making the most of each day together.
Rhonda:
We’re looking forward to taking a trip to Yellowstone, hopefully taking our grandkids and doing some hiking…
I don’t know that we would have been able to continue hiking with his chorea had it continued on without the medication…
I feel like that we’re actually like living the way we’ve always wanted to live now…
…by going on vacations and being outdoors a lot more and having grandkids…
And yeah, I can’t image life if he didn’t have the medication.
Ray:
It really is a deal-maker for me. It just helped so much.
Onscreen text:
WHAT COULD LONG-TERM RESULTS MEAN FOR YOUR PATIENTS WITH HD CHOREA?
Learn more at AUSTEDOhcp.com
Onscreen ISI scroll:
INDICATIONS AND USAGE
AUSTEDO XR and AUSTEDO are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO. Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, on AUSTEDOhcp.com.
Onscreen text:
This was an individual patient’s experience. Results may vary.
Reference: Data on file. Parsippany, NJ: Teva Neuroscience, Inc.

Metabolic Pathways
Visualizing the role of metabolic pathways in drug-drug interactions (DDIs).
Onscreen text:
CYP enzymes mediate the breakdown of active drug to inactive metabolites for clearance.
Together, CYP3A4/5 and CYP2D6 enzymes are involved in metabolizing ~75% drugs.
CYP enzymes play a key role in converting VMAT2 inhibitors into their active and inactive metabolites.
Certain drugs can act as strong CYP inhibitors or inducers, affecting the metabolism of coadministered medications…
…resulting in drug-drug interactions.
Inhibited metabolism
Inhibited metabolism results in elevated levels of active metabolites.
Elevated levels of active metabolites lead to increased drug potency and potential for adverse events.
Induced metabolism
Induced metabolism results in decreased levels of active metabolites.
Decreased levels of active metabolites diminish therapeutic effect.
For VMAT2 inhibitors, drug-drug interactions are determined by:
The CYP enzymes critical in metabolizing the VMAT2 inhibitor
AND
The metabolic profiles of the medications the patient is taking concomitantly (whether they are strong inhibitors or inducers)
To avoid drug-drug interactions associated with inhibited or induced metabolism, limiting VMAT2 inhibitor dose or avoiding use with strong inhibitors and inducers may be recommended.
When choosing a VMAT2 inhibitor, considering these drug-drug interactions is key.
VMAT2, vesicular monoamine transporter 2.
The Impact of HD Chorea on Daily Activities: Ray and Rhonda’s Story
See how AUSTEDO made a difference in physical functioning for Ray.
Onscreen text:
HD chorea
THE IMPACT OF HD CHOREA ON DAILY ACTIVITIES
RAY & RHONDA’S STORY
Disclaimer at bottom of screen:
This was an individual patient’s experience. Results may vary. Ray and Rhonda were compensated for their participation in this video.
Onscreen text:
HD, Huntington’s disease.
Rhonda:
As the movements progressed more and more, I was thinking about what equipment he would need for getting dressed or taking a shower. OR if he would need a wheelchair.
On-screen text:
99% of patients with HD chorea require assistance with self-care activities.
When Ray was diagnosed with HD in 2019, his chorea had already begun limiting his physical functioning.
Ray:
I first noticed my involuntary movements when we were working…and my hand would just shake. I was just thinking, what’s going on?
Rhonda:
I was noticing at night when we were sleeping…he would be kicking me or hitting…he was just flailing around, moving his arms and legs.
Onscreen text:
As Ray’s symptoms worsening, Ray and his wife, Rhonda, knew it was time to talk to his doctor.
Rhonda:
We spoke with the doctor about it, and we didn’t know if there was any options for dealing with the chorea or not.
But we did talk to her and let her know that this has been going on for quite some time, and just wondering if there was any help for that.
Onscreen text:
The physical functioning portion of the SF-36 questionnaire, a health-related quality of life scale, was used to assess the impact of Ray’s chorea on daily activities.
Bathing
Lifting/carrying groceries
Walking
Moderate to vigorous activities
Climbing stairs
Bending, kneeling, or stooping
SF-36, Short Form Health Survey.
Based on Ray’s symptoms and the impact his chorea had on his activities of daily living, he was prescribed AUSTEDO.
Onscreen Indication and Black Box Warning:
AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of tardive dyskinesia and for the treatment of chorea associated with Huntington's disease.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Onscreen text:
Patients taking AUSTEDO, like Ray, reported being better able to perform activities of daily living—with a significant 4.3-point difference in SF-36 physical functioning score in the pivotal trial.
Patients taking placebo had a decline in physical functioning as measured by SF-36
Rhonda:
He’s able to still do all the daily activities that one does during the day, all the ordinary things that sometimes the rest of us would take for granted.
Onscreen text:
More than 3 years later, Ray is still taking AUSTEDO at 48 mg/day. His chorea movements have remained minimal, and he’s better able to perform activities of daily living.
Disclaimer at bottom of screen:
Once-daily AUSTEDO XR is available.
Bioequivalence of once-daily AUSTEDO XR has been established with AUSTEDO BID based on pharmacokinetic profile studies. When two formulations are shown to be bioequivalence, they are considered to be therapeutically equivalent.
Rhonda:
Starting early on AUSTEDO has made a big difference in what we’re able to do now.
It keeps his movements at bay, keeps us hiking and walking and traveling and doing things that I don’t believe we would be able to do had he not got it started.
We’re actually like living the way we’ve always wanted to live now. I can’t imagine life if he didn’t have it.
Onscreen text:
Learn more about how AUSTEDO could make a different for your patients’ physical functioning at AUSTEDOhcp.com
Onscreen ISI scroll:
INDICATIONS AND USAGE
AUSTEDO XR and AUSTEDO are indicated in adults for the treatment of tardive dyskinesia and for the treatment of chorea associated with Huntington's disease.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity.
Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias. Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.
Onscreen text:
This was an individual patient’s experience. Results may vary.
References: 1. Claassen DO, DeCourcy J, Mellor J, Johnston C, Iyer RG. Impact of chorea on self-care activity, employment, and health-care resource use in patients with Huntington’s disease. J Health Econ Outcomes Res. 2021;8(1):99-105. 2. Data on file. Parsippany, NJ: Teva Neuroscience, Inc. 3. RAND Corporation. 36-Item Short Form Survey Instrument (SF-36). RAND Corporation. Accessed November 12, 2022. https://www.rand.org/health-care/surveys_tools/mos/36-item-short-form/survey-instrument.html

Easing the Burden for Patients and Caregivers: Nathan & Heather’s HD Chorea Journey
See how Nathan experienced movement reduction and easily switched from AUSTEDO to AUSTEDO XR.
Onscreen text:
EASING THE BURDEN FOR PATIENTS AND CAREGIVERS
NATHAN & HEATHER’S STORY
Disclaimer on bottom of screen:
This was an individual patient’s experience. Results may vary. Participants were compensated by Teva Pharmaceuticals.
[Blind footnote on bottom of screen]
HDc, Huntington’s disease chorea.
Heather:
I’m Heather. This is my husband, Nathan.
Nathan and I met through his sister, one of my best friends. We just adore each other. I don’t know any other way to put it.
Nathan:
I love her.
Heather:
He loves me.
Onscreen text:
Nathan and Heather were newlyweds when he was diagnosed with HD.
His chorea forced them to reconsider what their future together would look like.
Heather:
I knew nothing about Huntington’s disease until I met Nathan and his sister, and they would talk about their mom having the disease—it’s very scary.
Onscreen text:
Over time, Nathan’s symptoms worsened, and Heather soon became more than his wife—she became his caregiver.
Heather:
When Nathan first started needing my help, it was a bit more daunting. Our child was small, so I had them both depending on me.
He depended on me a lot…helping him walk, holding his hands. And I don’t mean the side by side holding hands, I mean I’m standing in front of him holding on to his arms to help him steady his gait.
Onscreen text:
In 2017, Nathan’s doctor started him on AUSTEDO.
Onscreen Indication and Black Box Warning:
AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington's disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Heather:
We started seeing a difference once Nathan started AUSTEDO—he was able to hold on to tools, pick up drink cans, play with our kids.
Chorea control gives me peace of mind for how I care for Nathan.
I don’t feel quite as much as a caregiver. I feel more like a wife and more a partner with my husband.
Onscreen text:
When once-daily AUSTEDO XR became available, Nathan’s doctor recommended the switch.
Heather:
The process of switching to AUSTEDO XR was so easy.
It was very easy to explain the dose schedule to friends and family—you only have to take AUSTEDO XR once a day, with or without food. It made life a whole lot easier.
We didn’t have to change any doses with his other medications.
We love that we can increase and decrease the dosages as needed.
Onscreen text:
Despite his HD progressing, Nathan continues to see chorea control more than 8 years after starting treatment.
Heather:
With disease progression, movement control gives us a better bond because we can sit and hold hands, we can enjoy watching a movie—we can enjoy things that we felt like we would never be able to enjoy again.
He can play video games and bond with our son.
We attribute AUSTEDO XR to helping him out considerably. I have my husband back a little bit again.
Onscreen text:
DISCOVER THE DIFFERENCE CHOREA MANAGEMENT CAN MAKE FOR PATIENTS AND THEIR CAREGIVERS AT AUSTEDOhcp.com
Onscreen ISI scroll
INDICATIONS AND USAGE
AUSTEDO XR and AUSTEDO are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.

Reclaiming Independence With Early Treatment: James & Sue's Story
See the difference early HD chorea treatment with AUSTEDO made for James and his wife and caregiver, Sue.
[title]
HDc
RECLAIMING INDEPENDENCE WITH EARLY TREATMENT
JAMES & SUE’S STORY
[description]
See the difference early HD chorea treatment with AUSTEDO made for James and his wife and caregiver, Sue.
Onscreen text:
HDc
RECLAIMING INDEPENDENCE WITH EARLY TREATMENT
JAMES & SUE’S STORY
Disclaimer on bottom of screen:
This was an individual patient’s experience. Results may vary. Participants were compensated by Teva Pharmaceuticals.
[Blind footnote on bottom of screen]
HDc, Huntington’s disease chorea.
James:
My name is James, and I’m 73 years old.
Sue:
My name is Sue, and I’m 75 years old.
James:
I met Sue in 1969 at the University of Rhode Island.
Sue:
We’ve been married 51 wonderful years.
Onscreen text:
James learned about HD through his family’s history.
James:
My mom had Huntington’s. She had a lot of severe symptoms.
I decided to get tested. I wanted to rule it out for our children.
Onscreen text:
James was diagnosed with HD early, before experiencing any symptoms.
But when he started noticing involuntary movements, he and Sue were prepared for their lives to change.
James:
When I first started experiencing chorea symptoms, I had a twitching in my toe.
It was a slow progression…the feet started to move, the legs and lower body.
When you’re out in public, people coming the other way on the sidewalk, they look at me a little more, because I’m stumbling or weaving around.
On-screen text with icons:
In a survey, most patients and caregivers stated that it is “very important” to manage chorea.*
[footnote]
*Survey included 115 patients with HD and caregivers (n=80 patients with HD; n=35 caregivers).
Patients’ and caregivers’ concerns include:
Loss of independence
Unpredictability/
Uncontrollability
Impact on family life
[disclaimer on cards]
No clinical trials have been conducted to demonstrate that treating HD chorea affects the outcomes listed above.
Sue:
I know there was gonna be changes, I know he’d lose some abilities to do some things.
James:
HD chorea affected my independence because of the movements in my fingers, they were gradually getting worse over time. Made it hard to hit a keyboard accurately. I was no longer able to type an email on a computer.
Super text:
Not long after James began experiencing movements, he and Sue talked to James’ doctor about chorea management.
He started taking AUSTEDO, and soon noticed that his movement improvements helped him regain some independence.
Onscreen Indication and Black Box Warning:
AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington's disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Sue:
Treating chorea early is important.
James:
I was proactive because of my mother.
It was pretty quickly that I noticed the results.
Sue:
Since James started with the AUSTEDO, I noticed a change in the movements of his legs which has been awesome and wonderful for both of us.
James:
AUSTEDO has helped me with several of my movements.
[disclaimer]
AUSTEDO XR has not been shown to affect the outcomes mentioned.
James:
I do feel more independent. I do feel better that people don’t notice.
Sue:
I would definitely recommend AUSTEDO for anyone I know whose loved one is suffering with chorea movements.
James:
Since I’ve decided to seek treatment, my life has been better overall.
Onscreen text:
EXPLORE THE DIFFERENCE CHOREA CONTROL WITH AUSTEDO XR CAN MAKE FOR YOUR PATIENTS AND THEIR CAREGIVERS AT AUSTEDOhcp.com
Onscreen ISI scroll:
INDICATIONS AND USAGE
AUSTEDO XR and AUSTEDO are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Learn more about AUSTEDO XR at AUSTEDOhcp.com.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.
For more videos, visit the YouTube page.
HD chorea: Patient Case Studies

Progressive HD Chorea: A Case
Study of Care and Treatment Presented by Danny Bega, MD
[Title]
PROGRESSIVE HD CHOREA:
A Case Study of Care and Treatment
Presented by Danny Bega, MD
[Description]
A Case Study of Care and Treatment: Explore the journey of a middle-aged woman with Huntington’s disease, to enhance understanding of HD chorea, with highlights of key clinical features, diagnosis, impact of symptoms, and treatment options.
Onscreen text:
Danny Bega, MD
Associate Professor of Neurology
at Northwestern University
Director of Huntington’s Disease
Clinic at Northwestern Medicine
Disclaimer at bottom of screen:
HD, Huntington’s disease.
Doctor Bega:
Hi, I’m Dr Danny Bega. I’m an Associate Professor of Neurology at Northwestern University, and the Director of the Huntington’s Disease Clinic at Northwestern Medicine.
On behalf of Teva Pharmaceuticals, I welcome you to this case study presentation, in which we will explore the journey of a middle-aged woman with Huntington’s disease, or HD. By exploring this patient's story, we aim to enhance understanding of HD chorea, and we will highlight key clinical features, diagnosis, impact of symptoms, and treatment options.
Onscreen text:
CHRISTY
Personal History
- 40- to 45-year old female
- Previously worked in an office. Stay-at-home mother
- Divorced; has 2 young adult children
Family History
- Her mother and grandmother had HD
- Her mother died in her 40s
Medical History
- Presymptomatic testing for HD reported 43 CAG repeats
- Has anxiety and is treating with an SSRI
- Chronically underweight
Doctor Bega:
Our patient—let’s call her Christy—is a woman in her early to mid-40s. Christy previously worked in an office but is currently a stay-at-home mother with 2 young adult children. She has a family history of HD, with her mother and grandmother both affected by this condition. Her mother passed away in her 40s.
Given her family history, Christy requested presymptomatic testing for HD, which revealed 43 CAG repeats in the HTT gene on chromosome 4. Additionally, she experiences anxiety and is on a selective serotonin reuptake inhibitor, or SSRI, to help manage it. She is also chronically underweight.
Onscreen text:
HD is caused by CAG repeat expansion (>36%) in the huntingtin gene, resulting in production of mutant huntingtin protein
There are ~200,000 patients at risk of developing HD in the United States
Doctor Bega:
HD is a hereditary, neurodegenerative disorder caused by an expansion of CAG repeats—36 or more—in the huntingtin gene. This genetic mutation leads to the production of a mutant huntingtin protein, which is central to the disease's pathophysiology. There are approximately 200,000 patients at risk of developing HD in the United States.
Onscreen text:
Cognitive
Behavioral/Emotional
Motor
Doctor Bega:
HD is characterized by a triad of cognitive, behavioral, and motor symptoms. Cognitive impairments often precede the diagnosis of HD by many years. Emotional and behavioral symptoms vary among individuals over the course of the disease.
Nearly 90% of patients ultimately develop chorea, which is characterized by continuous, involuntary, random, jerk-like movements that can occur early and continue throughout the course of the disease.
Onscreen text:
Cognitive
Cognitive impairments may emerge years before diagnosis of HE; however, cognitive decline is gradual
Behavioral/Emotional
Behavioral/emotional symptoms of HD are more variable and are associated with functional decline
Onscreen text:
Motor
Chorea, characterized by continuous, involuntary, random, jerk-like movements is the most prominent motor symptom and appears early in the course of the disease
Motor
Chorea, characterized by continuous, involuntary, random, jerk-like movements is the most prominent motor symptom and appears early in the courses of the disease Nearly 90% of patients with HD develop chorea, with varying severity over the course of the disease
Onscreen text:
CHOREA, IRRESPECTIVE OF SEVERITY, CAN IMPACT PATIENTS’ DAILY ACTIVITIES AND FUNCTIONING
Mobility
Getting dressed/washed
Preparing/cooking meals
Taking medication
Eating
No clinical studies have been conducted to demonstrate that treating HC chorea affects the outcomes shown here
Doctor Bega:
Chorea, regardless of the level of severity, can impact patients’ daily activities and functioning. Patients with chorea may have trouble with mobility and may require aids such as a cane or a walking stick, may have trouble getting washed and dressed without help, often need help preparing and cooking meals, and can have difficulty taking medication or eating without assistance.
Onscreen text:
Care partners’ perception of the most impactful symptoms of HD (N=701)
Care partners rate chorea as the most impactful symptom
Doctor Bega:
Chorea has been reported as the most impactful symptom of HD by care partners.
Onscreen text:
Presentation at the HDSA Center of Excellence
- Development of movements in fingers and toes prompted the visit
- Patient was accompanied by her aunt who had concerns related to driving and financial decision-making
- Patient reported being completely independent and able to manage all her activities
Due to lack of awareness, discussion about concerns with driving and financial decision-making were upsetting for Christy
HDSA, Huntington’s Disease Society of America.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bega:
Christy first presented to the HDSA Center of Excellence complaining of movements in her fingers and toes. Her aunt expressed concerns about Christy's ability to drive safely and manage her finances effectively. However, Christy reported being completely independent and capable of managing all her activities without assistance. This lack of self-awareness made discussions about her driving and financial decisions particularly upsetting for Christy.
Onscreen text:
Anosognosia, or lack of insight, can occur throughout the course of HD and may impact care and the role of care partners
The care partner’s perspective is important when determining the impact of symptoms
Doctor Bega:
Patients with HD often exhibit anosognosia, or lack of insight, which can result in their inability to recognize or accurately assess their own symptoms. Therefore, it may be helpful to get the care partner’s perspective when determining the impact of symptoms.
Onscreen text:
CHRISTY
Cognitive
Behavioral/Emotional
Motor
Cognitive
MoCA score was 23/30; patient did not report any cognitive concerns
MoCA, Montreal Cognitive Assessment.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bega:
Though Christy did not report any cognitive concerns, her Montreal Cognitive Assessment, or MoCA, revealed mild cognitive impairment with a score of 23 out of 30.
Emotionally and behaviorally, her anxiety is well-managed with an SSRI. She has no issues with sleep.
A motor exam identified mild finger and toe fidgeting, dysarthria, impaired saccadic eye movements, and choreoathetosis in distal extremities, with choreiform movements noted while walking. Despite these symptoms, she expressed no interest in treating her chorea at that time.
Christy was referred to a multidisciplinary care team for additional support and was advised to schedule follow-up visits every 4 months.
Onscreen text:
Behavioral/Emotional
- Anxiety (controlled by an SSRI)
- No sleep issues
Motor
- Mild finger and toe fidgeting
- Dysarthria
- Impaired saccadic eye movements
- Choreoathetosis in distal extremities, with choreiform movements while walking (no falls reported)
Onscreen text:
At this visit, patient was not interested in treating HD chorea. Patent was referred to a multidisciplinary care team and was advised to follow up every 4 months
Onscreen text:
Huntington’s Disease Society of America Centers of Excellence
Dietitian
Genetic counselor
Neurologist
Psychiatrist
Nurse
Physical therapist/PM&R specialist
Occupational therapist
Psychologist
Speech therapist
Social worker
PM&R, physical medicine and rehabilitation.
Doctor Bega:
As part of Christy’s multidisciplinary support, she was given a referral to a physical medicine and rehabilitation specialist, who provided tailored exercise recommendations aimed at enhancing gait and balance, thereby reducing the risk of falls. A dietitian created a nutritional management plan focusing on addressing Christy's chronic low weight. A speech therapist performed a swallowing evaluation. Additionally, a social worker worked with Christy to navigate discussions around giving up driving to improve her safety, offered assistance with financial management and organizing medical appointments, helped her understand disability benefits and long-term care planning options, and connected her with support groups. Finally, a genetic counselor provided education and insights about genetic risks to help Christy in discussing testing options for at-risk family members.
Onscreen text:
CHRISTY
CASE STUDY: FOLLOW-UP 1 YEAR LATER
Clinical Assessment
- Facial, orolingual, and trunk chorea observed
- Limb chorea was more pronounced
- Patient was unable to complete a tandem walk
Personal Status
- Had increased anxiety
- Was no longer driving
- Was living with her aunt who assisted with medications, doctor appointments, finance, and other daily activities
- Aunt expressed concern about poor financial decisions, impulsivity, and excessive substance abuse
Observations and testing confirmed that the patient’s chorea symptoms were impacting balance, ambulation, and social interactions
UHDRS TMS = 18
TMS, Total Motor Score; UHDRS, Unified Huntington’s Disease Rating Scale.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bega:
At a follow-up visit a year later, facial, orolingual, and trunk chorea were observed, limb chorea was more pronounced, and Christy could not complete a tandem walk. Her Total Motor Score, or TMS, was 18. The TMS is one part of the Unified Huntington's Disease Rating Scale, or UHDRS, which offers an overall assessment of various domains of motor function including chorea, dystonia, tongue protrusion, eye movements, gait, and balance.
Christy also reported increased anxiety and was no longer driving, per ongoing counseling from her social worker. She was living with her aunt who assisted with medication management, doctor appointments, finances, and daily activities. Separately, her aunt expressed concerns about Christy’s poor financial decisions, impulsivity, and excessive substance abuse, all of which appeared to reflect the increasing impact of her disease.
At this point, both observation and testing confirmed that Christy’s chorea symptoms were affecting her balance, ambulation, and social interactions.
Onscreen text:
- How is chorea affecting daily activities and social interactions?
- Does it take longer to accomplish a task at home or at work?
- As a care partner, how much time are you spending to help with daily activities?
- Has chorea led to stigma for you or your family?
- Is chorea contributing to concerns with balance/falls and overall safety (swallowing, dysphagia, burns)?
Onscreen text:
Do you find that people have trouble following what you say?
Doctor Bega:
Understanding the impact of HD chorea on patients and care partners is crucial when making treatment decisions. To assess impact, it's important to engage with both the patient and their care partner by asking questions that require thoughtful responses vs replying with a simple yes or no. Asking about the patient’s quality of life, successful completion of daily activities, and physical or psychosocial challenges may help patients or care partners convey the true impact of mild or early-stage chorea and help prioritize treatment decisions. One question I often ask is whether they have noticed any changes or impacts on their speech, or if others are finding it difficult to follow what they're saying, because speech articulation can be a sign of chorea in the orobuccal-lingual area.
Onscreen text:
CHRISTY
- A shared decision was made to treat chorea with AUSTEDO, a VMAT2 inhibitor approved for the treatment of HC chorea
- To address Christy’s increased anxiety, she was referred to a psychiatrist who increased the dosage of her SSRI
VMAT2, vascular monoamine transporter 2.
Patient profile based upon a real patient case. Actor portrayal.
INDICATIONS AND USAGE
AUSTEDO® XR and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.

Chorea in Huntington's Disease: A Patient's Journey from Diagnosis to Treatment Presented by Bisena
Bulica, DO
CHOREA IN HUNTINGTON’S DISEASE:
A Patient’s Journey from Diagnosis to Treatment
Presented by Bisena Bulica, DO
Onscreen text:
Bisena Bulica, DO
Director and Senior Staff Neurologist
Henry Ford Health
West Bloomfield, MI
Disclaimer at bottom of screen:
Dr. Bulica has received compensation from Teva to give this presentation.
Doctor Bulica:
Hi, I’m Dr Bisena Bulica. I’m the director of the Huntington’s Disease Center of Excellence at Henry Ford Health in West Bloomfield, Michigan. Welcome to this case study presentation, sponsored by Teva Neuroscience. I have received compensation from Teva to give this presentation. Today, we will explore the journey of a patient living with Huntington’s disease, or HD. The goal is to deepen your understanding of HD chorea by examining its main clinical features, the impact on patients and their care partners, and to review treatment options currently available.
Onscreen text:
CAROL
Personal History
- 50- to 55-year-old female
- Healthcare professional
- Divorced with 2 children aged 18-25 years
Family History
- Carol’s father, paternal aunt, and grandmother had HD
Footnotes:
HD, Huntington’s disease.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bulica:
Our patient, Carol, is a woman in her mid-50s. She works in healthcare, is divorced, and lives with her 2 grown children. She has a family history of HD. Her father, paternal aunt, and grandmother all had the condition.
Initially, Carol visited her neurologist with concerns about her balance. Given her family history of HD, she underwent immediate genetic testing that showed 41 CAG repeats in the huntingtin gene on chromosome 4.
Carol has a history of depression, which is well managed with a selective serotonin reuptake inhibitor, or SSRI. Carol has also been diagnosed with hypertension and fibromyalgia.
Onscreen text:
CAROL
Medical History
- Presented to a general neurologist with concerns about balance
- Genetic testing for HD reported 41 CAG repeats
- Comorbidities include depression, hypertension, and fibromyalgia
Treatment History
- Treated with an SSRI for depression
Footnotes:
CAG, cytosine, adenine, guanine.
Patient profile based upon a real patient case. Actor portrayal.
Onscreen text:
HD is caused by a CAG repeat expansion (≥36) in the huntingtin gene, resulting in production of mutant huntingtin protein
There are ~200,000 patients at risk of developing HD in the United States
Doctor Bulica:
HD is an inherited neurodegenerative condition caused by an expansion of 36 or more CAG repeats in the huntingtin gene. This genetic mutation results in the creation of a mutant huntingtin protein, which is central to the disease’s development. In the United States, there are approximately 41,000 patients with HD and approximately 200,000 individuals are at risk of developing HD.
With 41 CAG repeats, Carol’s HD diagnosis was confirmed. To address her balance, she was advised to begin physical therapy. At this time, no additional treatments were suggested.
Onscreen text:
CAROL
- With 41 CAG repeats, Carol’s HD diagnosis was confirmed
- To address her balance, she was advised to begin physical therapy
- At this time, no additional treatments were suggested
Onscreen text:
Cognitive
Emotional/Behavioral
Motor
Doctor Bulica:
HD is characterized by a triad of cognitive, behavioral, and motor symptoms. Cognitive impairments often precede the diagnosis of HD by many years. Emotional and behavioral symptoms may vary among individuals over the course of the disease.
Over 90% of patients ultimately develop chorea, which is characterized by continuous, involuntary, random, jerk-like movements that can occur early on and continue throughout the course of the disease.
Onscreen text:
Cognitive
Cognitive impairments may emerge years before diagnosis of HD; however, cognitive decline is gradual
Onscreen text:
Emotional/Behavioral
Emotional/behavioral symptoms of HD are more variable and are associated with functional decline
Onscreen text:
Motor
Chorea, characterized by continuous, involuntary, random, jerk-like movements, is the most prominent motor symptom and appears early in the course of the disease
Nearly 90% of patients with HD develop chorea, with varying severity over the course of the disease
Onscreen text:
CAROL
Presentation 1 Year Post HD Diagnosis
- Clinical examination revealed chorea symptoms had worsened
- Involuntary movements in the feet disrupted gait, leading to falls
- Tongue movements impaired speech and chewing abilities
- Involuntary movements of the lips and jaws were socially embarrassing
- Reduced dexterity was making Carol dependent on her children, who also expressed concerns about her driving
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bulica:
A year later, Carol returned to her neurologist as her chorea symptoms had worsened. The involuntary movements in her feet were disrupting her gait and causing falls. Tongue movements led to impaired speech and difficulty chewing. Social embarrassment was another concern, as the involuntary movements in her lips and jaws were attracting unwanted attention and reactions from both her family members and strangers.
At home, Carol was becoming more dependent on her children who were helping her manage her finances and assisting with household chores. She noticed that everyday tasks were taking longer to complete, and that she required additional time to dress and prepare whenever she needed to go out.
Her children raised concerns about her driving, describing it as jerky and unsafe.
Onscreen text:
CHOREA, IRRESPECTIVE OF SEVERITY, CAN IMPACT PATIENTS’ DAILY ACTIVITIES AND FUNCTIONING
Mobility
Getting dressed/washed
Preparing/cooking meals
Taking medication
Eating
No clinical studies have been conducted to demonstrate that treating HC chorea affects the outcomes shown here.
Doctor Bulica:
Chorea, irrespective of its severity, can significantly affect a patient’s everyday activities and overall functioning. Those with chorea might struggle with mobility, often necessitating the use of assisted devices like a cane or a walking stick. They may also find it challenging to bathe and dress independently, frequently requiring assistance. Often, tasks like preparing and cooking meals can be difficult, and some patients may need help with taking their medications as prescribed.
Onscreen text:
Care partners’ perception of the most impactful symptoms of HD (N=701)
Care partners rate chorea as the most impactful symptom
Doctor Bulica:
Care partners often cite chorea as the most impactful symptom of HD.
Onscreen text:
CAROL
Treatment
- The general neurologist initiated treatment with tetrabenazine 25 mg BID
Footnotes:
BID, twice daily.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bulica:
Due to the impact of chorea symptoms on Carol’s life, she was prescribed treatment with tetrabenazine 25 mg twice daily. At her follow-up appointment 1 month later, there was mild improvement in her chorea symptoms, but Carol was experiencing sedation. To manage this, her dose of tetrabenazine was reduced to 12.5 mg twice daily. Carol continued on this lower dose for 6 months, but it was not an adequate dose to manage her chorea symptoms. Furthermore, even on this reduced dose, she continued to experience daytime somnolence. Her tetrabenazine dosage was increased back to 25 mg administered once daily at night, to reduce the impact of sedation throughout the day. Carol was referred to the Huntington’s Disease Society of America Center of Excellence, or HDSA COE, for further management.
Onscreen text:
CAROL
Follow-up at 1 month
- Very mild improvement in chorea symptoms
- Sedation was reported as a side effect
To address the side effect, tetrabenazine dose was reduced to 12.5 mg BID
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
>Onscreen text:
CAROL
Follow-up at 6 months
- No significant improvement in chorea symptoms was seen
- Carol reported experiencing daytime somnolence
Dose of tetrabenazine was increased to 25 mg once daily and Carol was referred to the HDSA Center of Excellence
Footnotes:
HDSA, Huntington’s Disease Society of America.
Patient profile based upon a real patient case. Actor portrayal.
Onscreen text:
Dietician
Genetic counselor
Neurologist
Psychiatrist
Nurse
Physical therapist/PM&R specialist
Occupational therapist
Psychologist
Speech therapist
Social worker
The COE provides access to multidisciplinary clinical care and services through a geographically diverse network of local and/or regional clinical centers
Footnote:
PM&R, physical medicine and rehabilitation.
Doctor Bulica:
The HDSA COEs provide a multidisciplinary team approach to care for patients with HD. Patients benefit from expert neurologists, who specialize in movement disorders such as Huntington’s disease, psychiatrists, social workers, physical therapists, occupational therapists, genetic counselors, and other professionals who have expertise working with patients and families affected by HD.
Onscreen text:
CAROL
Clinical Assessment
- Carol’s patient and treatment history were obtained
- UHDRS was used to document her TMS and TMC scores
- TMS = 36
- TMC = 12
Carol was offered to switch from tetrabenazine 25 mg BID to once-daily AUSTEDO XR
Footnotes:
UHDRS, Unified Huntington’s Disease Rating Scale; TMC, total motor chorea; TMS, total motor score.
Patient profile based upon a real patient case. Actor portrayal.
INDICATIONS AND USAGE AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Doctor Bulica:
At the HDSA COE, Carol’s medical and treatment history was thoroughly examined. The Unified Huntington’s Disease Rating Scale, or UHDRS, was used to record her total motor score, or TMS, and her total maximal chorea, or TMC, score. The TMS is a 15-item assessment tool designed to evaluate motor symptoms in individuals with HD, examining chorea, dystonia, parkinsonism, motor performance, oculomotor function, and balance. The ratings can range from 0 to 124.
The TMC score, which is a component of the UHDRS TMS, assesses movements across seven areas of the body—including the face, mouth, trunk, arms, and legs—on a scale from 0 to 4. The total TMC score ranges from 0 to 28, with lower scores indicating less severe chorea.
In Carol’s assessment, her UHDRS TMS score was 36 and TMC score was 12.
Following all assessments, Carol was presented with the option to switch from taking tetrabenazine 25 mg once daily to a once-daily dose of AUSTEDO XR.
Onscreen text:
ONCE-DAILY AUSTEDO XR®
FOR CHOREA ASSOCIATED WITH HD
Onscreen text:
INDICATIONS AND USAGE AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of chorea associated with
Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Doctor Bulica:
AUSTEDO XR is a one pill, once-daily treatment option for adults with chorea associated with HD. Careful consideration should be taken when prescribing due to the increased risk of depression and suicidality in patients.
Onscreen text:
TMC Score Over Time (ITT Population, N=90)
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
- TMC score reduced by 4.4 points vs 1.9 points with placebo (P<0.0001)*
- 90% of patients saw improvement in their movements vs baseline†
- 33% of patients achieved a ≥6-point improvement compared to 2% treated with placebo
~40% reduction in chorea severity vs baseline at Week 12 (~40 mg/day)‡
Footnotes:
*Mean total dose.
†Versus maintenance therapy (average of Weeks 9 and 12).
‡Patients saw a ≥1-point reduction in TMC score by Week 12.
BID, twice daily; ITT, intention-to-treat; SEM, standard error of the mean.
Doctor Bulica:
Clinical trials supporting the FDA approval of AUSTEDO were carried out using the twice-daily formulation. The FIRST-HD study was a randomized, double-blind, placebo-controlled trial aimed at evaluating the efficacy and safety of AUSTEDO. The treatment period consisted of an 8-week dose titration phase followed by a 4-week maintenance phase.
The main measure of efficacy in this trial was the treatment effect of AUSTEDO compared with placebo, as determined by changes in the TMC score from baseline through the maintenance period.
After 12 weeks, the TMC scores showed an improvement of 4.4 points with AUSTEDO, compared with a 1.9-point improvement in the placebo group, reflecting an approximate 40% reduction in chorea severity. Notably, 90% of patients experienced improvement in their movements compared with baseline, with 33% achieving a 6-point or greater improvement, in contrast to just 2% of those treated with placebo.
Onscreen text:
LS Mean Change in TMS from baseline maintenance therapy
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
LS, least square.
Doctor Bulica:
The study also looked at the changes in the UHDRS TMS, which showed greater than 2 times reduction in total motor impairment with AUSTEDO vs placebo.
Onscreen Text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Walking
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Doctor Bulica:
Improvements in physical functioning were assessed using the 36-Item Short Form Health Survey, or SF-36 Physical Functioning Score.
Patients taking AUSTEDO achieved a greater improvement in SF-36 physical functioning scores at 12 weeks vs placebo and reported improvement in their ability to perform daily activities like walking, bathing, dressing, lifting, moderate to vigorous activities, climbing stairs, and bending, kneeling, or stooping.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Bathing
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Dressing
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Lifting
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Moderate to vigorous activities
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Climbing stairs
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like
Bending, kneeling, or stooping
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
Most common AEs in FIRST-HD (reported in ≥4% of patients on AUSTEDO)
AE, AUSTEDO (n=45) %, Placebo (n=45) %
Somnolence 11, 4
Diarrhea 9, 0
Dry mouth 9, 7
Fatigue 9, 4
Urinary tract infection 7, 2
Insomnia 7, 4
Irritability 7, 13
Dizziness 4, 9
Anxiety 4, 2
Constipation 4, 2
Contusion 4, 2
Fall 4, 9
Depression/agitated depression 4, 7
Adverse reactions with AUSTEDO XR are expected to be similar to AUSTEDO BID.
Footnote:
AE, adverse event.
Doctor Bulica:
The most common adverse events in the AUSTEDO group were somnolence, diarrhea, dry mouth, and fatigue. Once patients were titrated to their maintenance dose, these adverse events were no longer reported. 2% of participants taking AUSTEDO withdrew from the study due to side effects. Adverse events of special interest such as parkinsonism, akathisia, restlessness, and suicidal ideation were not reported during the titration phase. During the maintenance phase, they were reported in 4% or fewer patients across both arms.
Anxiety was reported in 4% or fewer patients in both arms during the titration and maintenance phases.
Onscreen text:
TMC Score Reduction Maintained Through 3 Years
Patients in the ARC-HD study received the AUSTEDO BID formulation.
TMC reductions were seen in both cohorts at Week 8 and maintained through Week 145
Doctor Bulica:
Now let’s look at the results from the open-label extension study that evaluated the long-term safety of AUSTEDO. In this study, which was not placebo-controlled, 119 people with HD chorea received AUSTEDO for about 3 years. This study showed that AUSTEDO maintained chorea improvement of HD over the long term. TMC score reduction was maintained for about 3 years and there were no new safety concerns in this open-label study.
Onscreen text:
CAROL
Tetrabenazine to AUSTEDO XR
- Carol was interested in trying treatment with once-daily AUSTEDO XR
- She was switched overnight from 25 mg/day tetrabenazine to 12 mg/day AUSTEDO XR
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Doctor Bulica:
Carol was eager to try a different treatment regimen with AUSTEDO XR, and she was switched overnight from 25 mg/day tetrabenazine to 12 mg/day AUSTEDO XR.
Onscreen text:
Recommended Initial Dose Following Switch at ~50% of Daily Tetrabenazine Dose
Current Daily Tetrabenazine Dose
12.5 mg
25 mg
37.5 mg
50 mg
62.5 mg
75 mg
87.5 mg
100 mg
Initial AUSTEDO XR Dose
6 mg QD
12 mg QD
18 mg QD
24 mg QD
30 mg QD
36 mg QD
42 mg QD
48 mg QD
Resume recommended titration schedule after tetrabenazine switch.
- Patients may initiate AUSTEDO XR on the day following discontinuation of tetrabenazine
- After patients are switched to AUSTEDO XR, the dose may be adjusted at weekly intervals
Footnotes:
QD, once daily.
AUSTEDO XR® (deutetrabenazine) extended-release tables/AUSTEDO® current Prescribing Information. Parsippany, NJ: Teva Neuroscience, Inc.
Doctor Bulica:
Transitioning from tetrabenazine to AUSTEDO XR is straightforward. Treatment with AUSTEDO XR can begin the day after stopping tetrabenazine. It is recommended to start AUSTEDO XR at a dose approximately 50% of the total daily dose of tetrabenazine being taken.
Onscreen text:
ONE PILL, ONCE DAILY WITH OR WITHOUT FOOD
TITRATE WITH 4-WEEK KIT + Rx
WEEK 1, 12 mg
WEEK 2, 18 mg
WEEK 3, 24 mg
WEEK 4, 30 mg
4-week Titration Kit
available for all new patient starts (sample or prescription)
WEEK 5, 36 mg
WEEK 6, 42 mg
WEEK 7, 48 mg
Average daily dose in clinical trials was >36 mg/day
Most patients were between 36 and 48 mg/day at the end of the long-term study
AUSTEDO XR should be swallowed whole. Tablets should not be chewed, crushed, or broken.
Image shown is not actual 4-week Titration Kit. Tablets not shown to actual size.
Doctor Bulica:
AUSTEDO XR offers flexible dosing that effectively and tolerably manages chorea symptoms. This once-daily medication begins with a 12 mg/day dose, increasing by 6 mg/day each week until an effective and tolerable dose is reached. The maximum dose of AUSTEDO XR is 48 mg/day, with the average dose in clinical trials being over 36 mg daily.
Patients should swallow AUSTEDO XR tablets whole, without chewing, crushing, or breaking them.
To initiate treatment with AUSTEDO XR, a 4-week Titration Kit is available, either as a sample provided directly to the patients or through a prescription from a specialty pharmacy of their choice.
Onscreen text:
CAROL
Treatment
- Carol initiated AUSTEDO XR at 12 mg/day, which was increased weekly by 6 mg/day to a maintenance dose of 36 mg/day
- She continues to take an SSRI for depression
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Dr Bulica:
Carol started AUSTEDO XR at a 12 mg dosage, which was increased weekly by 6 mg/day to a maintenance dose of 36 mg/day.
In addition to AUSTEDO XR, Carol continues to take an SSRI to manage her depression.
As part of the multidisciplinary support, Carol was referred to a genetic counselor who offered education and insights about the genetic risks for her children. They also discussed the ramifications of pursuing genetic testing for them in the future. Additionally, a physical therapist provided exercise recommendations and monitored improvements in her gait and balance. A speech therapist conducted periodic evaluations of her swallowing ability, and a social worker ensured Carol and her family were informed about available resources in the community and at the national level. She was also connected with support groups in her area.
Onscreen text:
CAROL CONTINUES TO RECEIVE SUPPORT FROM HER MULTIDISCIPLINARY TEAM
Dietician
Genetic counselor
Neurologist
Psychiatrist
Nurse
Physical therapist/PM&R specialist
Occupational therapist
Psychologist
Speech therapist
Social worker
Following Carol’s positive outcome with AUSTEDO XR, she is now an active member of different support groups
Onscreen text:
CAROL
Outcome
- Improved dexterity while using computer, writing, or using kitchen utensils
- Overall improved balance as reported in her physical therapy assessments
UHDRS Assessment
- TMS = 31
- TMC = 7
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Dr. Bulica:
Treatment with AUSTEDO XR led to a reduction in the severity of Carol’s involuntary movements. Carol noticed improved dexterity when using her computer, writing, or handling kitchen utensils. Her physical therapist also noted an overall improvement in her balance. Additionally, her TMS improved from 36 to 31, and her TMC score improved from 12 to 7.
Her children reported that she required less help with household chores. She could now handle working in the kitchen and preparing her own meals with minimal assistance from them.
Onscreen text:
Significant chorea control in placebo-controlled studies
Dr. Bulica:
To summarize, AUSTEDO has demonstrated efficacy in controlling chorea symptoms in patients with HD, as shown in placebo-controlled studies, with improvement maintained for approximately 3 years in an open-label extension study. AUSTEDO has a demonstrated safety profile. Studies have also shown significant improvements in physical functioning, leading to a reduction in disability. AUSTEDO XR is designed with flexibility to provide effective and tolerable symptom control, along with convenient once-daily dosing.
Onscreen text:
Sustained improvement through ~3 years (OLE study)
Footnote:
OLE, open-label extension.
Onscreen text:
Demonstrated safety profile
Onscreen text:
Proven disability reduction backed by significant improvement in physical functioning
Onscreen text:
Flexibility for effective and tolerable control
Onscreen text:
Easy and convenient once-daily dosing
Doctor Bulica:
Thank you for viewing this case study presentation. I trust it has offered you valuable insights that you can apply in your clinical practice to improve the care of patients with HD.
Onscreen ISI scroll and voiceover:
INDICATIONS AND USAGE
AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.

From Out of Nowhere: The Onset
and Impact of HD Chorea Presented
by Yasar Torres-Yaghi, MD
[Title]
FROM OUT OF NOWHERE:
The Onset and Impact of HD Chorea
Presented by Yasar Torres-Yaghi, MD
Onscreen text:
Yasar Torres-Yaghi, MD
Director, Georgetown University Hospital
Assistant Professor, Georgetown University School of Medicine
Disclaimer at bottom of screen:
Dr Torres-Yaghi has received compensation from Teva to give this presentation.
Doctor Torres-Yaghi:
Hi, I’m Dr Torres-Yaghi. I’m the Director of the Parkinsonism and Dementia Clinic at Georgetown University Hospital and an Assistant Professor at Georgetown University School of Medicine. Welcome to this case study presentation, sponsored by Teva Neuroscience, Inc. I have received compensation from Teva to give this presentation. Today, we will explore the diagnostic journey of a patient with Huntington’s disease, or HD, with no prior family history. We will also examine the clinical features of chorea associated with HD, recognize its impact on patients and their families, and review current treatment options.
Onscreen text:
DAVID
Personal History
- Mid-60s male
- Works in construction, maintenance, and repair
- Married with 2 children aged 30 to 36 years
Medical History
- Depression
- 1 to 2 years of progressive, involuntary movements affecting gait and balance
Treatment History
- SSRI for depression
Footnotes:
HD, Huntington’s disease; SSRI, selective serotonin reuptake inhibitor.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
Our patient, David, is a man in his mid-60s. He works in construction, maintenance, and repair. He is married with 2 children who are in their early-to-mid 30s. David has a history of depression, which is managed with a selective serotonin reuptake inhibitor, or SSRI.
About 1 to 2 years ago, David started experiencing involuntary movements in his limbs that were affecting his gait and balance. Since they were not impacting his daily activities, he did not pay much attention.
Onscreen text:
DAVID
PROGRESSION OF SYMPTOMS OVER THE LAST YEAR
Involuntary Movements
- Increased movements in limbs resulting in falls
- Development of movements in the head and neck
Emotional/Behavioral Symptoms
- Worsened depression, irritability, and social withdrawal
- Onset of repetitive behaviors
Cognitive Symptoms
- Memory loss
- Problems with planning
- Difficulty paying attention
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
However, over the past year, the symptoms continued to progress, resulting in falls and the development of additional movements in his head and neck.
David also started showing new emotional and behavioral symptoms. His wife noted that his depression had worsened, and she also noted irritability, social withdrawal, and the development of compulsive behaviors, such as frequently checking locks and whether the stove was turned off. He also began to experience cognitive challenges, including memory loss, problems with planning, and difficulty paying attention.
Onscreen text:
DAVID
Impact of Symptoms
- Impaired thinking
- Decreased motivation at work
- Strained relationship with colleagues
- Social isolation from friends and family
David’s wife and children encouraged him to visit his primary care physician, who recognized the need for further evaluation and referred him to a neurologist
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
These symptoms not only disrupted David’s daily life but also started affecting his work performance. His cognitive and psychiatric challenges led to impaired thinking and decreased motivation, resulting in fewer completed projects, missed deadlines, and strained relationships with colleagues. At home, he was less interested in pursuing his hobbies, seeing friends, and was spending less time with his children.
Due to these changes, David’s wife and children encouraged him to visit his primary care physician, who recognized the need for further evaluation and referred him to a neurologist.
Onscreen text:
DAVID
Clinical Examination by a Neurologist
- Clinical examination revealed
- Generalized chorea
- Impaired saccadic eye movements
- Unsteady gait
- MoCA Score of 24/30
Treatment
- To address his psychiatric symptoms, David was started on an antipsychotic
His neurologist suspected HD and referred him to an HDSA COE
Footnotes:
HDSA COE, Huntington’s Disease Society of America Center of Excellence; MoCA, Montreal Cognitive Assessment.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
Clinical examination at his neurologist’s office revealed generalized chorea, impaired saccadic eye movements, and an unsteady gait. His Montreal Cognitive Assessment, or MoCA, revealed mild cognitive impairment with a score of 24 out of 30. To address his psychiatric symptoms, David was prescribed an antipsychotic. Based on David’s symptoms, his neurologist suspected HD, despite a lack of family history for the condition. David was referred to a Huntington’s Disease Society of America Center of Excellence for further evaluation.
Onscreen text:
HD is caused by a CAG repeat expansion (≥36) in the huntingtin gene, resulting in production of mutant huntingtin protein
There are ~41,000 patients with HD and ~200,000 patients at risk of developing HD in the United States
Footnote:
CAG, cytosine-adenine-guanine.
Doctor Torres-Yaghi:
HD is a hereditary, neurodegenerative disorder caused by an expansion of CAG repeats—36 or more—in the huntingtin gene. This genetic mutation leads to the production of a mutant huntingtin protein, which is central to the disease's pathophysiology. In the United States, there are approximately 41,000 patients with HD, and approximately 200,000 individuals are at risk of developing HD.
Onscreen text:
Cognitive
Emotional/Behavioral
Motor
Doctor Torres-Yaghi:
HD is characterized by a triad of cognitive, behavioral, and motor symptoms. Cognitive impairments often precede the diagnosis of HD by many years. Emotional and behavioral symptoms may vary among individuals over the course of the disease. Over 90% of patients ultimately develop chorea, which is characterized by continuous, involuntary, random, jerk-like movements that can occur early on and continue throughout the course of the disease.
Onscreen text:
Cognitive
Cognitive impairments may emerge years before diagnosis of HD; however, cognitive decline is gradual
Onscreen text:
Emotional/Behavioral
Emotional/behavioral symptoms of HD are more variable and are associated with functional decline
Onscreen text:
Motor
Chorea, characterized by continuous, involuntary, random, jerk-like movements, is the most prominent motor symptom and appears early in the course of the disease
NEARLY 90% of patients with HD develop chorea, with varying severity over the course of the disease
Onscreen text:
CHOREA, IRRESPECTIVE OF SEVERITY, CAN IMPACT PATIENTS’ DAILY ACTIVITIES AND FUNCTIONING
Mobility
Getting dressed/washed
Preparing/cooking meals
Taking medication
Eating
Footnote:
No clinical studies have been conducted to demonstrate that treating HD chorea affects the outcomes shown here.
Doctor Torres-Yaghi:
Chorea, irrespective of its severity, can significantly affect a patient’s everyday activities and functioning. Those with chorea might struggle with mobility, often necessitating the use of assistive devices like a cane or walking stick. They may also find it challenging to bathe and dress independently, frequently requiring assistance. Often, tasks like preparing and cooking meals can be difficult, and some patients may need help with taking their medications as prescribed. Care partners consider chorea as the most impactful symptom of HD.
Onscreen text:
Care partners’ perception of the most impactful symptoms of HD (N=701)
30% Chorea
24% Cognitive decline
15% Depression/apathy
15% Speech and swallowing issues
Care partners rate chorea as the most impactful symptom
Onscreen text:
DAVID
Clinical Assessment at the HDSA COE
- Involuntary movements evaluated by a movement disorder specialist
- UHDRS was used to document his TMS and TMC scores
- TMS=25
- TMC=19
Genetic testing for HD was recommended
Footnotes:
TMC, Total Maximal Chorea; TMS, total motor score; UHDRS, Unified Huntington’s Disease Rating Scale.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
At the HDSA COE, David’s involuntary movements were evaluated by a movement disorder specialist, and his medical and treatment history was thoroughly examined. The Unified Huntington’s Disease Rating Scale, or UHDRS, was used to record his total motor score, or TMS, and total maximal chorea, or TMC, score. The TMS is a 15-item assessment tool designed to evaluate motor symptoms in individuals with HD, examining chorea, dystonia, parkinsonism, motor performance, oculomotor function, and balance. The ratings can range from 0 to 124.
The TMC score, which is a component of the UHDRS TMS, assesses movements across 7 areas of the body—including the face, mouth, trunk, arms, and legs—on a scale from 0 to 4. The total TMC score ranges from 0 to 28, with lower scores indicating less severe chorea.
David’s UHDRS TMS score was 25, and his TMC score was 19.
David was referred for genetic testing to confirm the diagnosis.
Onscreen text:
Patient Goes to the Doctor
Chorea
Known Family History of HD; No Known Family History of HD; Medical and Medication History
Genetic Testing
Positive; Negative
Diagnosis; Other Tests and Investigations
Doctor Torres-Yaghi:
For patients with a known family history of HD, genetic testing is a clear and immediate step to confirm the diagnosis once symptoms appear. Similarly, for patients without a family history of HD, genetic testing should be conducted if other potential causes of involuntary movements are ruled out through medical and medication history. If genetic testing results are negative, other tests and imaging investigations should be conducted. Using this approach helps patients and their families by preventing delays in treatment.
Onscreen text:
DAVID
Genetic testing for HD revealed 42 CAG repeats in the HTT gene
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
David’s genetic testing revealed 42 CAG repeats in the HTT gene on chromosome 4, confirming the diagnosis of HD. Sometimes, patients with HD may lack a family history, especially in those with late onset disease. In those cases, relatives or family members with HD may not have exhibited symptoms during their lifetime or had mild symptoms that went unrecognized prior to dying of other causes.
Onscreen text:
DAVID
- To address David’s chorea symptoms, a decision was made to treat with AUSTEDO XR, a VMAT2 inhibitor approved for the treatment of HD chorea
Footnotes:
VMAT2, vesicular monoamine transporter 2.
Patient profile based upon a real patient case. Actor portrayal.
INDICATIONS AND USAGE
AUSTEDO XR® and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Please see additional Important Safety Information at the end of this video.
Doctor Torres-Yaghi:
To address his chorea symptoms, David was prescribed AUSTEDO XR.
Onscreen text:
ONCE-DAILY AUSTEDO XR®
FOR CHOREA ASSOCIATED WITH HD
Doctor Torres-Yaghi:
AUSTEDO XR, a VMAT2 inhibitor, is a one pill, once-daily treatment option for adults with chorea associated with HD. Careful consideration should be taken when prescribing due to the increased risk of depression and suicidality in patients.
Onscreen text:
FIRST-HD PRIMARY ENDPOINT: CHANGE IN TMC SCORE FROM BASELINE TO MAINTENANCE THERAPY (N=90, ITT)
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
- TMC score reduced by 4.4 points vs 1.9 points with placebo (P<0.0001)*
- 90% of patients saw improvement in their movements vs baseline†
- 33% of patients achieved a ≥6-point improvement compared with 2% treated with placebo
~40% reduction in chorea severity vs baseline at Week 12 (~40 mg/day)‡
Footnotes:
*Versus maintenance therapy (average of Weeks 9 and 12).
†Patients saw a ≥1-point reduction in TMC score by Week 12.
‡Mean total dose.
BID, twice daily; ITT, intention-to-treat.
Doctor Torres-Yaghi:
The FIRST-HD study, which supported the FDA approval of the AUSTEDO BID formulation, was a randomized, double-blind, placebo-controlled trial aimed at evaluating the efficacy and safety of AUSTEDO. The treatment period consisted of an 8-week dose titration phase followed by a 4-week maintenance phase.
The main measure of efficacy in this trial was the changes in the TMC score from baseline through the maintenance period of AUSTEDO compared with placebo.
After 12 weeks, the TMC scores showed an improvement of 4.4 points with AUSTEDO, compared with a 1.9-point improvement in the placebo group, reflecting an approximate 40% reduction in chorea severity in the treatment group.
90% of patients experienced improvement in their movements from baseline. 33% achieved a 6-point or greater improvement, compared to 2% of those treated with placebo.
Onscreen text:
LS MEAN CHANGE IN TMS FROM BASELINE MAINTENANCE THERAPY
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
LS, least square.
Doctor Torres-Yaghi:
The study also assessed the changes in the UHDRS TMS, which showed greater than 2 times reduction in total motor impairment with AUSTEDO vs placebo.
Onscreen Text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Walking
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Doctor Torres-Yaghi:
Improvements in physical functioning were assessed using the 36-Item Short Form Health Survey, or SF-36 Physical Functioning Score.
Patients taking AUSTEDO achieved a greater improvement in SF-36 physical functioning scores at 12 weeks vs placebo and reported improvement in their ability to perform daily activities like walking, bathing, dressing, lifting, moderate to vigorous activities, climbing stairs, and bending, kneeling, or stooping.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Bathing
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Dressing
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Lifting
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Moderate to vigorous activities
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Climbing stairs
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
ONLY VMAT2 INHIBITOR WITH PATIENT-REPORTED IMPROVEMENT IN ABILITY TO PERFORM ACTIVITIES OF DAILY LIVING
Patients reported improvement in the SF-36 Physical Functioning Score at 12 weeks vs placebo in activities like Bending, kneeling, or stooping
AUSTEDO XR/AUSTEDO does not cure the cause of the involuntary movements, and it does not treat the other symptoms of HD, such as problems with thinking or emotions.
Patients in the FIRST-HD study received the AUSTEDO BID formulation.
Footnote:
SF-36, 36-Item Short Form Health Survey.
Onscreen text:
MOST COMMON AEs IN FIRST-HD (REPORTED IN ≥4% OF PATIENTS ON AUSTEDO)
AE, AUSTEDO (n=45) %, Placebo (n=45) %
Somnolence 11, 4
Diarrhea 9, 0
Dry mouth 9, 7
Fatigue 9, 4
Urinary tract infection 7, 2
Insomnia 7, 4
Irritability 7, 13
Dizziness 4, 9
Anxiety 4, 2
Constipation 4, 2
Contusion 4, 2
Fall 4, 9
Depression/agitated depression 4, 7
Adverse reactions with AUSTEDO XR are expected to be similar to AUSTEDO BID.
Footnote:
AE, adverse event.
Doctor Torres-Yaghi:
The most common adverse events in the AUSTEDO group were somnolence, dry mouth, diarrhea, and fatigue. Once patients were titrated to their maintenance dose, these adverse events were no longer reported. 2% of participants taking AUSTEDO withdrew from the study due to side effects. Adverse events of special interest, parkinsonism, akathisia, restlessness, and suicidal ideation were not reported during the titration phase. During the maintenance phase, they were reported in 4% or fewer of patients in both arms.
Anxiety was reported in 4% or fewer patients in both arms during the titration and maintenance phases.
Onscreen text:
TMC SCORE REDUCTION MAINTAINED THROUGH 3 YEARS
Patients in the ARC-HD study received the AUSTEDO BID formulation.
TMC reductions were seen in both cohorts at Week 8 and maintained through Week 145
Doctor Torres-Yaghi:
In the open-label extension study, which was not placebo-controlled, 119 people with HD chorea received AUSTEDO for about 3 years. This study showed that AUSTEDO maintained chorea improvement over the long term. TMC score reduction was maintained for about 3 years, and there were no new safety concerns in this open-label study.
Onscreen text:
ONE PILL, ONCE DAILY WITH OR WITHOUT FOOD
TITRATE WITH 4-WEEK KIT + Rx
WEEK 1, 12 mg
WEEK 2, 18 mg
WEEK 3, 24 mg
WEEK 4, 30 mg
4-week Titration Kit
available for all new patient starts (sample or prescription)
WEEK 5, 36 mg
WEEK 6, 42 mg
WEEK 7, 48 mg
Average daily dose in clinical trials was >36 mg/day
Flexibility to titrate weekly until effective and tolerable symptom control is reached (48 mg max dose)
Most patients were between 36 and 48 mg/day at the end of the long-term study
AUSTEDO XR should be swallowed whole. Tablets should not be chewed, crushed, or broken.
Image shown is not actual 4-week Titration Kit. Tablets not shown to actual size.
Doctor Torres-Yaghi:
AUSTEDO XR offers flexible dosing that effectively and tolerably manages chorea symptoms. This once-daily medication begins with a 12 mg/day dose, increasing by 6 mg/day each week until an effective and tolerable dose is reached. The maximum dose of AUSTEDO XR is 48 mg/day, with the average dose in clinical trials being over 36 mg daily.
Patients should swallow AUSTEDO XR tablets whole, without chewing, crushing, or breaking them.
To initiate treatment with AUSTEDO XR, a 4-week Titration Kit is available, either as a sample provided directly to the patients or through a prescription.
Onscreen text:
DAVID
Treatment
- Initiated on AUSTEDO XR at 12 mg/day, which was increased weekly by 6 mg/day to a maintenance dose of 36 mg/day
Footnote:
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
David started AUSTEDO XR at 12 mg/day, and the dose was increased by 6 mg/day weekly to a maintenance dose of 36 mg/day.
His SSRI was increased to address his depression and OCD symptoms.
For his cognitive symptoms, a cholinesterase inhibitor was prescribed.
Onscreen text:
DAVID
- SSRI increased to address depression and OCD symptoms
- Cholinesterase inhibitor initiated to address cognitive impairment
Footnotes:
OCD, obsessive-compulsive disorder.
Patient profile based upon a real patient case. Actor portrayal.
Onscreen text:
DAVID CONTINUES TO RECEIVE SUPPORT FROM HIS MULTIDISCIPLINARY TEAM
Dietitian
Genetic counselor
Neurologist
Psychiatrist
Nurse
Physical therapist/PM&R specialist
Occupational therapist
Psychologist
Speech therapist
Social worker
Footnote:
PM&R, Physical Medicine and Rehabilitation.
Doctor Torres-Yaghi:
David receives ongoing support from a multidisciplinary team, not only for the motor, cognitive, and emotional and behavioral symptoms of HD, but also to address the functional impact on him and his family. Physical therapy improved his balance and reduced the risk of falls; occupational therapy helped with both gross and fine motor functioning, and speech therapy helped improve his executive functioning. In addition, his children will receive genetic counseling to learn about the implications of presymptomatic testing, and also support to help manage the emotional and psychological challenges that the family might face.
Onscreen text:
DAVID
Outcome
- Patient rated symptoms as “very much improved” on the PGIC scale
- David was able to return to work
UHDRS Assessment
- TMS=18
- TMC=13
Footnotes:
PGIC, Patient Global Impression of Change.
Patient profile based upon a real patient case. Actor portrayal.
Doctor Torres-Yaghi:
Treatment with AUSTEDO XR led to an overall improvement in David’s chorea and physical functioning. On the Patient Global Impressions of Change Scale, David rated his symptoms as very much improved. His TMS improved from 25 to 18, and his TMC score improved from 19 to 13. David has now returned to work.
Onscreen text:
Significant chorea control in placebo-controlled studies
Doctor Torres-Yaghi:
To summarize, AUSTEDO has demonstrated efficacy in controlling chorea symptoms in patients with HD, as shown in placebo-controlled studies, with improvement maintained for approximately 3 years in an open-label extension study. AUSTEDO has a demonstrated safety profile. Studies have also shown significant improvements in physical functioning, leading to a reduction in disability. AUSTEDO XR is designed with flexibility to provide effective and tolerable symptom control, along with convenient once-daily dosing.
Onscreen text:
Sustained improvement through ~3 years (OLE study)
Footnote:
OLE, open-label extension.
Onscreen text:
Demonstrated safety profile
Onscreen text:
Proven disability reduction backed by significant improvement in physical functioning
Onscreen text:
Flexibility for effective and tolerable control
Onscreen text:
Easy and convenient once-daily dosing
Doctor Torres-Yaghi:
Thank you for participating in this case study presentation. I hope it has offered you valuable insights to assist in diagnosing HD and enhancing the care of patients experiencing chorea associated with HD.
INDICATIONS AND USAGE
AUSTEDO® XR and AUSTEDO® are indicated in adults for the treatment of chorea associated with Huntington’s disease and for the treatment of tardive dyskinesia.
IMPORTANT SAFETY INFORMATION
Depression and Suicidality in Patients with Huntington’s Disease:
AUSTEDO XR and AUSTEDO can increase the risk of depression and suicidal thoughts and behavior (suicidality) in patients with Huntington’s disease. Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior. Inform patients, their caregivers, and families of the risk of depression and suicidality and instruct them to report behaviors of concern promptly to the treating physician. Exercise caution when treating patients with a history of depression or prior suicide attempts or ideation. AUSTEDO XR and AUSTEDO are contraindicated in patients who are suicidal, and in patients with untreated or inadequately treated depression.
Contraindications: AUSTEDO XR and AUSTEDO are contraindicated in patients with Huntington’s disease who are suicidal, or have untreated or inadequately treated depression. AUSTEDO XR and AUSTEDO are also contraindicated in: patients with hepatic impairment; patients taking reserpine or within 20 days of discontinuing reserpine; patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of discontinuing MAOI therapy; and patients taking tetrabenazine or valbenazine.
Clinical Worsening and Adverse Events in Patients with Huntington’s Disease: AUSTEDO XR and AUSTEDO may cause a worsening in mood, cognition, rigidity, and functional capacity. Prescribers should periodically re-evaluate the need for AUSTEDO XR or AUSTEDO in their patients by assessing the effect on chorea and possible adverse effects.
QTc Prolongation: AUSTEDO XR and AUSTEDO may prolong the QT interval, but the degree of QT prolongation is not clinically significant when AUSTEDO XR or AUSTEDO is administered within the recommended dosage range. AUSTEDO XR and AUSTEDO should be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias.
Neuroleptic Malignant Syndrome (NMS), a potentially fatal symptom complex reported in association with drugs that reduce dopaminergic transmission, has been observed in patients receiving tetrabenazine. The risk may be increased by concomitant use of dopamine antagonists or antipsychotics. The management of NMS should include immediate discontinuation of AUSTEDO XR and AUSTEDO; intensive symptomatic treatment and medical monitoring; and treatment of any concomitant serious medical problems.
Akathisia, Agitation, and Restlessness: AUSTEDO XR and AUSTEDO may increase the risk of akathisia, agitation, and restlessness. The risk of akathisia may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops akathisia, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Parkinsonism: AUSTEDO XR and AUSTEDO may cause parkinsonism in patients with Huntington’s disease or tardive dyskinesia. Parkinsonism has also been observed with other VMAT2 inhibitors. The risk of parkinsonism may be increased by concomitant use of dopamine antagonists or antipsychotics. If a patient develops parkinsonism, the AUSTEDO XR or AUSTEDO dose should be reduced; some patients may require discontinuation of therapy.
Sedation and Somnolence: Sedation is a common dose-limiting adverse reaction of AUSTEDO XR and AUSTEDO. Patients should not perform activities requiring mental alertness, such as operating a motor vehicle or hazardous machinery, until they are on a maintenance dose of AUSTEDO XR or AUSTEDO and know how the drug affects them. Concomitant use of alcohol or other sedating drugs may have additive effects and worsen sedation and somnolence.
Hyperprolactinemia: Tetrabenazine elevates serum prolactin concentrations in humans. If there is a clinical suspicion of symptomatic hyperprolactinemia, appropriate laboratory testing should be done and consideration should be given to discontinuation of AUSTEDO XR and AUSTEDO.
Binding to Melanin-Containing Tissues: Deutetrabenazine or its metabolites bind to melanin-containing tissues and could accumulate in these tissues over time. Prescribers should be aware of the possibility of long-term ophthalmologic effects.
Common Adverse Reactions: The most common adverse reactions for AUSTEDO (>8% and greater than placebo) in a controlled clinical study in patients with Huntington’s disease were somnolence, diarrhea, dry mouth, and fatigue. The most common adverse reactions for AUSTEDO (4% and greater than placebo) in controlled clinical studies in patients with tardive dyskinesia were nasopharyngitis and insomnia. Adverse reactions with AUSTEDO XR extended-release tablets are expected to be similar to AUSTEDO tablets.
Please see full Prescribing Information, including Boxed Warning, at AUSTEDOhcp.com.
SF-36, Short Form (36) Health Survey; VMAT2, vesicular monoamine transporter 2.
PointClickCare® is a registered trademark of PointClickCare Technologies Inc.
REFERENCE: 1. Solmi M, Pigato G, Kane JM, Correll CU. Treatment of tardive dyskinesia with VMAT-2 inhibitors: a systematic review and meta-analysis of randomized controlled trials. Drug Des Devel Ther. 2018;12:1215-1238.